Abstract
Background
Chemotherapy-induced peripheral neuropathy (CIPN) is a troublesome chronic symptom that has no proven pharmacologic treatment. The purpose of this double-blind randomized placebo-controlled trial was to evaluate a novel compounded topical gel for this problem.
Methods
Patients with CIPN were randomized to baclofen 10 mg, amitriptyline HCL 40 mg, and ketamine 20 mg in a pluronic lecithin organogel (BAK-PLO) versus placebo (PLO) to determine its effect on numbness, tingling, pain, and function. The primary endpoint was the baseline-adjusted sensory subscale of the EORTC QLQ-CIPN20, at 4 weeks.
Results
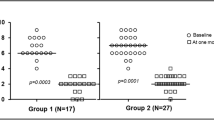
Data in 208 patients reveal a trend for improvement that is greater in the BAK-PLO arm over placebo in both the sensory (p = 0.053) and motor subscales (p = 0.021). The greatest improvements were related to the symptoms of tingling, cramping, and shooting/burning pain in the hands as well as difficulty in holding a pen. There were no undesirable toxicities associated with the BAK-PLO and no evidence of systemic toxicity.
Conclusion
Topical treatment with BAK-PLO appears to somewhat improve symptoms of CIPN. This topical gel was well tolerated, without evident systemic toxicity. Further research is needed with increased doses to better clarify the clinical role of this treatment in CIPN.



Similar content being viewed by others
References
Cavaletti G, Bogliun G, Marzorati L et al (1995) Peripheral neurotoxicity of Taxol in patients previously treated with cisplatin. Cancer 75:1141–1150
Cavaletti G, Fabbrica D, Minoia C et al (1998) Carboplatin toxic effects on the peripheral nervous system of the rat. Ann Oncol 9:443–447
Ocean JA, Vahdat LT (2004) Chemotherapy-induced peripheral neuropathy: pathogenesis and emerging therapies. Support Care Cancer 12:619–625
Pace A, Bove L, Aloe A et al (1997) Paclitaxel neurotoxicity: clinical and neurophysiological study of 23 patients. Ital J Neurol Sci 18:73–79
Rowinsky EK, Chaudry V, Cornblath DR et al (1993) Neurotoxicity of taxol. J Natl Cancer Inst Monogr 15:107–115
Verstappen C, Heimans JJ, Hockman K et al (2003) Neurotoxic complications of chemotherapy in patients with cancer. Drugs 63(15):1549–1563
Windebank AJ (1999) Chemotherapeutic neuropathy. Curr Opin Neurol 5:565–571
Campana WM, Eskeland N, Calcutt NA et al (1998) Prosaptide prevents paclitaxel neurotoxicity. Neurotoxicology 19:237–244
Cliffer KD, Siuciak JA, Carson SR et al (1998) Physiological characterization of taxol-induced large-fiber sensory neuropathy in the rat. Ann Neurol 43:46–55
Guastalla JP, Pujade-Lauraine E, Weber B (1998) Efficacy and safety of paclitaxel and carboplatin combination in patients with previously treated advanced ovarian carcinoma. Ann Oncol 9(9):37–43
Polomano RC, Bennett GJ (2001) Chemotherapy-evoked peripheral neuropathy. Pain Medicine 2:8–14
Pujude-Lauraine E, Guastalla JP, Weber B et al (1997) Efficacy and safety of the combination of paclitaxel/carboplatin in patients with previously treated advanced ovarian carcinoma: a multicenter French Groupe des investigateurs Nationauz pour l'etude des cancers ovariens phase II study. Semin Oncol 24:S15-30–S15-35
Quasthoff S, Hartung HP (2002) Chemotherapy induced peripheral neuropathy. J Neurol 249:9–17
Envig AI, Wiernik PH, Wadler S et al (1998) Phase I study of paclitaxel (taxol) and granulocyte colony stimulating factor (g-csf) in patients with unresectable malignany. Invest New Drugs 16:29–36
LoMonaco M, Milone M, Batocchi AP et al (1992) Cisplatin neuropathy: clinical course and neurophysiologic findings. J Neurol 239:199
Nikcevich DA, Grothey A, Sloan JA, Kugler JW, Silberstein PT, Dentchev T, Wender DB, Novotny PJ, Windschitl HE, Loprinzi CL (2008) Effect of intravenous calcium and magnesium (IV CaMg) on oxaliplatin-induced sensory neurotoxicity (sNT) in adjuvant colon cancer: results of the phase III placebo-controlled, double-blind NCCTG trial N04C7. J Clin Oncol 26:(abstr 4009)
Ahmad M, Goucke CR (2002) Management strategies for the treatment of neuropathic pain in the elderly. Drugs Aging 19(12):929–945
Argoff CE, Katz N, Backonja M (2004) Treatment of postherpetic neuralgia:a review of therapeutic options. J Pain Symptom Manage 28(4):396–411
Jensen PG, Larson JR (2001) Management of painful diabetic neuropathy. Drugs Aging 18(10):737–749
Hammack JE et al (2002) Phase III evaluation of nortriptyline for alleviation of symptoms of cis-platinum-induced peripheral neuropathy. Pain 98(1–2):195–203
Rao RD, Michalak JC, Sloan JA et al (2007) Efficacy of gabapentin in the management of chemotherapy-induced peripheral neuropathy: a phase 3 randomized, double-blind, placebo-controlled, crossover trial (N00C3). Cancer 110(9):2110–2118
Rao RD, Flynn PJ, Sloan J et al (2008) Efficacy of lamotrigine in the management of chemotherapy-induced peripheral neuropathy: a phase 3 randomized, double blind, placebo controlled NCCTG trial, N01C3. Cancer 112(21):2802–2808
Moore DH, Donnelly J, McGuire WP et al (2003) Limited access trial using amifostine for protection against cisplatin- and three-hour paclitaxel-induced neurotoxicity: a phase ii study of the Gynecologic Oncology Group. J Clin Oncol 21(22):4207–4213
Vahdat L, Papadopoulos K, Lange D et al (2001) Reduction of paclitaxel-induced peripheral neuropathy with glutamine. Clin Cancer Res 7:1192–1197
Baron (2006) Mechanisms of disease: neuropathic pain, a clinical perspective. Nat Clin Pract Neurol 2(2):95–106
Jones M (2000) Chronic neuropathic pain: pharmacological interventions in the new millennium. Int J Pharm Comp 4(1):6–15
Sawynok J (2005) Topical analgesics in neuropathic pain. Curr Pharm Des 11(23):2995–3004
Sawynok J (2003) Topical and peripherally acting analgesics. Pharmacol Rev 55:1–20
Lynch ME, Clark AJ, Sawynok J (2003) A pilot study examining topical amitriptyline, ketamine and a combination of both in the treatment of neuropathic pain. Clin J Pain 19:323–328
Lynch ME, Clark AJ, Sawynok J, Sullivan MJL (2005) Topical 2% amitriptyline and 1% ketamine in neuropathic pain syndromes. Anesthesiology 103(1):140–146
Lynch ME, Clark AJ, Sawynok J, Sullivan MJ (2005) Topical amitriptyline and ketamine in neurotopical amitriptyline and ketamine in neuropathic pain syndromes: an open label study. J Pain 6(10):644–649
Lockhart E (2004) Topical combination of amitriptyline and ketamine for post herpetic neuralgia. J Pain 5:S182
Everton D, Bhagwat D, Damask M (2007) An open-label pharmacokinetic study in humans of a 4% amitriptyline, 2% ketamine topical cream. J Pain 8(4 suppl 1):S48
Ceschel G, Bergamante V, Maffei P et al (2005) Solubility and transdermal permeation properties of dehydroepiandrosterone cyclodextrin complex from hydrophilic and lipophilic vehicles. Drug Delivery 12:275–280
Franek M, Vaculin S, Rokyta R (2004) GABAB receptor agonist baclofen has non-specific antinociceptive effect in the model of peripheral neuropathy in the rat. Pysiol Res 53:351–355
Smith GD, Harrison SM, Birch PJ et al (1994) Increased sensitivity to the antinociceptive activity of ± baclofen in an animal model of chronic neuropathic, but not chronic inflammatory hyperalgesia. Neuropharmacology 33:1103–1108
Gammaitoni A, Gallagher RM, Welz-Bosna M (2000) Topical ketamine gel: possible role in treating neuropathic pain. Pain Medicine 1(1):97–100
Pocock SJ, Simon R (1975) Sequential treatment assignment with balancing for prognostic factors in the controlled clinical trial. Biometrics 31:103–115
Postma TJ, Aaronson NK, Heimans JJ, EORTC Quality of Life Group et al (2005) The development of an EORTC quality of life questionnaire to assess chemotherapy-induced peripheral neuropathy: the QLQ-CIPN20. Eur J Cancer 41(8):1135–1139
McNair DM, Loor M, Droppleman LF (2003) Profile of mood states manual. Multi-Health Systems, New York
Cleeland CS, Ryan KM (1994) Pain assessment: global use of the brief pain inventory. Ann Acad Med Singapore 23(2):129–138
Sloan JA, O'Fallon JR, Suman VJ et al (1998) Incorporating quality of life measurement into oncology clinical trials. Proc Am Stat Assoc 1998:282–287
Grunberg SM, Groshen S, Steingass S (1996) Comparison of conditional quality of life terminology and visual analogue scale measurements. Qual Life Res 5:65–72
Gudex C, Dolan P, Kind P (1996) Health state valuations from the general public using the visual analogue scale. Qual Life Res 5:521–531
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was conducted as a collaborative trial of the North Central Cancer Treatment Group and Mayo Clinic and was supported in part by Public Health Service grants CA-25224, CA-37404, CA-63848, CA-35195, CA-37417, CA-35448, CA-35267, CA-63849, CA-35113, CA-35103, CA-35415, CA-35431, and CA124477. The content is solely the responsibility of the authors and does not necessarily represent the views of the National Cancer Institute or the National Institute of Health.
Additional participating institutions include the following: Cedar Rapids Oncology Program CCOP, Cedar Rapids, IA 52403, USA (Martin Wiesenfeld, MD); Geisinger Clinic and Medical Center CCOP, Danville, PA 17822, USA (Albert M. Bernath, Jr., M.D.); Rapid City Regional Hospital, Inc, Rapid City, SD 57701, USA (Richard Charles Tenglin, M.D.); Sioux Community Cancer Consortium, Sioux Falls, SD 57105, USA (Loren K. Tschetter, M.D.); Toledo Community Hospital Oncology Program (Paul L. Schaefer, M.D.); Metro-Minnesota Community Clinical Oncology Program, St. Louis Park, MN 55416, USA (Patrick J. Flynn, M.D.); Mayo Clinic, Scottsdale, AZ 85259-5404, USA (Tom R. Fitch, M.D.); and Hematology/Oncology Centers of the Northern Rockies, Billings, MT 59101, USA (Benjamin Marchello, M.D.).
Rights and permissions
About this article
Cite this article
Barton, D.L., Wos, E.J., Qin, R. et al. A double-blind, placebo-controlled trial of a topical treatment for chemotherapy-induced peripheral neuropathy: NCCTG trial N06CA. Support Care Cancer 19, 833–841 (2011). https://doi.org/10.1007/s00520-010-0911-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-010-0911-0